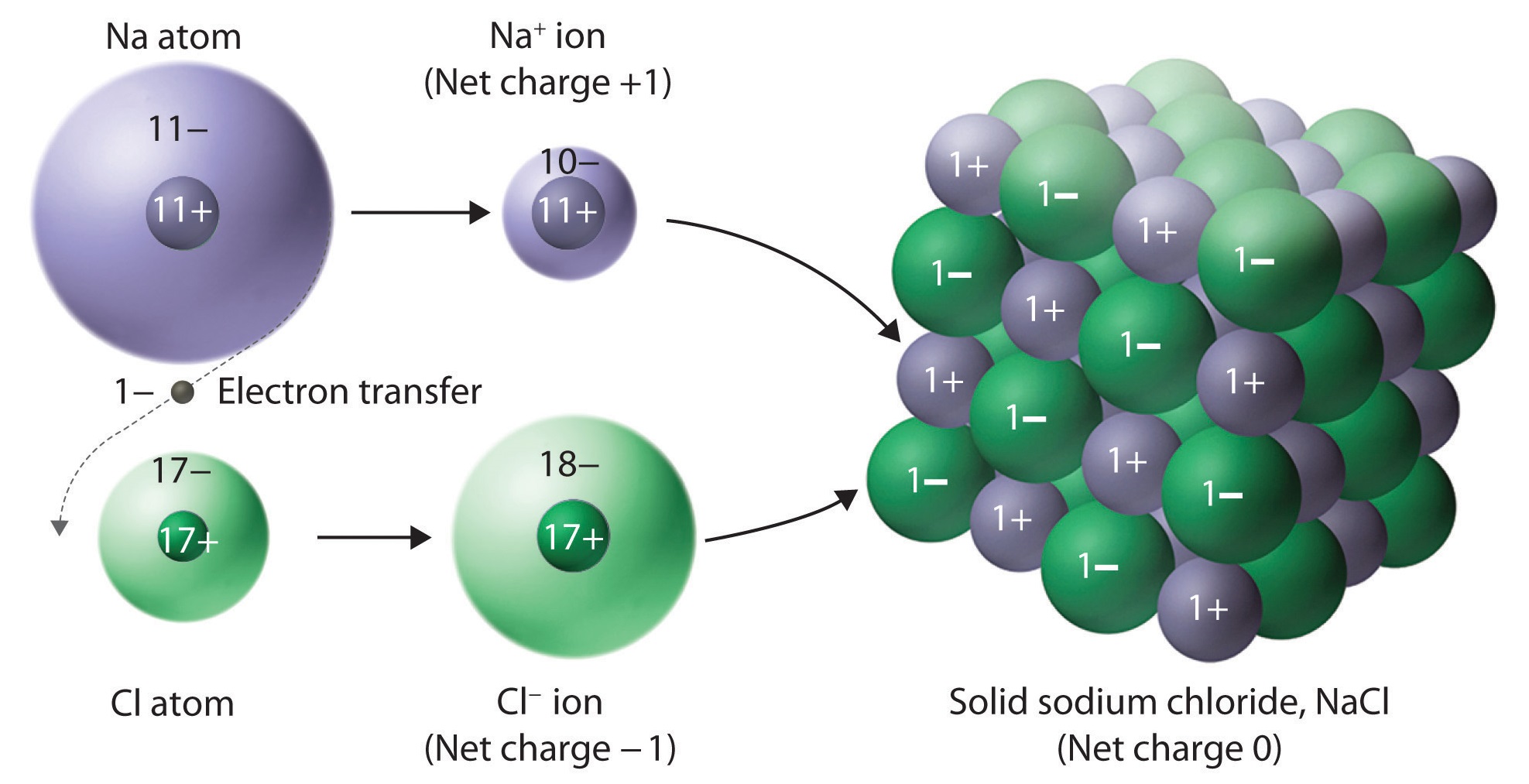
Does Boron Not Form Ionic Compounds . with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a metallic. boron can form ions but there is some fine print. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. to say that boron forms only covalent compounds is an oversimplification. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. It can be involved in metallic or even predominantly ionic. You won't get monatomic cations like the metals below it.
from chem.libretexts.org
with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a metallic. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. to say that boron forms only covalent compounds is an oversimplification. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. It can be involved in metallic or even predominantly ionic. You won't get monatomic cations like the metals below it. boron can form ions but there is some fine print.
Ionic Solids Chemistry LibreTexts
Does Boron Not Form Ionic Compounds You won't get monatomic cations like the metals below it. It can be involved in metallic or even predominantly ionic. to say that boron forms only covalent compounds is an oversimplification. boron can form ions but there is some fine print. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. You won't get monatomic cations like the metals below it. with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a metallic.
From www.doubtnut.com
Why boron forms electron deficient compounds? Does Boron Not Form Ionic Compounds You won't get monatomic cations like the metals below it. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. It can be involved in metallic or even predominantly ionic. with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a. Does Boron Not Form Ionic Compounds.
From medium.com
What is Boron? Periodic Table Elements Does Boron Not Form Ionic Compounds boron can form ions but there is some fine print. with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a metallic. It can be involved in metallic or even predominantly ionic. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with. Does Boron Not Form Ionic Compounds.
From www.britannica.com
boron Properties, Uses, & Facts Britannica Does Boron Not Form Ionic Compounds to say that boron forms only covalent compounds is an oversimplification. boron can form ions but there is some fine print. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. You won't get monatomic cations like the metals below it. Boron is found in nature on. Does Boron Not Form Ionic Compounds.
From borates.today
Boron Electron Valence Borates Today Does Boron Not Form Ionic Compounds You won't get monatomic cations like the metals below it. boron can form ions but there is some fine print. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. It can be involved in metallic or even predominantly ionic. to say that boron forms only covalent. Does Boron Not Form Ionic Compounds.
From www.youtube.com
SOME IMPORTANT COMPOUNDS OF BORON YouTube Does Boron Not Form Ionic Compounds purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. You won't get monatomic cations like the metals below it. It can be involved in metallic or even predominantly ionic. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other. Does Boron Not Form Ionic Compounds.
From www.britannica.com
crystal Types of bonds Britannica Does Boron Not Form Ionic Compounds Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. It can be involved in metallic or even predominantly ionic. with its high ionization energy, low electron affinity,. Does Boron Not Form Ionic Compounds.
From www.shalom-education.com
Properties of Ionic Compounds GCSE Chemistry Revision Does Boron Not Form Ionic Compounds purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. It can be involved in metallic or even predominantly ionic. to say that boron forms only covalent compounds is an oversimplification. with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does. Does Boron Not Form Ionic Compounds.
From learninglibraryhaney.z19.web.core.windows.net
Explain The Formation Of Ions Does Boron Not Form Ionic Compounds with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a metallic. boron can form ions but there is some fine print. You won't get monatomic cations like the metals below it. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with. Does Boron Not Form Ionic Compounds.
From collegedunia.com
Uses of Boron and Aluminium Properties and Solved Examples Does Boron Not Form Ionic Compounds with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a metallic. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree. Does Boron Not Form Ionic Compounds.
From periodictable.com
Sample of the element Boron in the Periodic Table Does Boron Not Form Ionic Compounds You won't get monatomic cations like the metals below it. to say that boron forms only covalent compounds is an oversimplification. boron can form ions but there is some fine print. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. purely ionic bonding cannot exist, as. Does Boron Not Form Ionic Compounds.
From mavink.com
Covalent Bond Types Does Boron Not Form Ionic Compounds You won't get monatomic cations like the metals below it. to say that boron forms only covalent compounds is an oversimplification. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated. Does Boron Not Form Ionic Compounds.
From www.sliderbase.com
Ionic Bonding Presentation Chemistry Does Boron Not Form Ionic Compounds with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a metallic. boron can form ions but there is some fine print. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. to say that boron forms only covalent. Does Boron Not Form Ionic Compounds.
From askfilo.com
Which of the following compounds of boron does not exist in the free form.. Does Boron Not Form Ionic Compounds It can be involved in metallic or even predominantly ionic. to say that boron forms only covalent compounds is an oversimplification. You won't get monatomic cations like the metals below it. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. boron can form ions but there is. Does Boron Not Form Ionic Compounds.
From schematicheymann5m.z4.web.core.windows.net
Boron Electron Dot Diagram Does Boron Not Form Ionic Compounds It can be involved in metallic or even predominantly ionic. boron can form ions but there is some fine print. with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a metallic. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with. Does Boron Not Form Ionic Compounds.
From www.britannica.com
Ionic bond Definition, Properties, Examples, & Facts Britannica Does Boron Not Form Ionic Compounds It can be involved in metallic or even predominantly ionic. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. to say that boron forms only covalent compounds is an oversimplification. with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does. Does Boron Not Form Ionic Compounds.
From www.nagwa.com
Question Video Explaining the Covalent Bonding in Borane Nagwa Does Boron Not Form Ionic Compounds It can be involved in metallic or even predominantly ionic. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. purely ionic bonding cannot exist, as the proximity of the entities involved in the bonding allows some degree of sharing. You won't get monatomic cations like the metals below. Does Boron Not Form Ionic Compounds.
From techiescientist.com
Boron Bohr Model Diagram, Steps To Draw Techiescientist Does Boron Not Form Ionic Compounds You won't get monatomic cations like the metals below it. Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. It can be involved in metallic or even predominantly ionic. to say that boron forms only covalent compounds is an oversimplification. with its high ionization energy, low electron. Does Boron Not Form Ionic Compounds.
From chem.libretexts.org
Ionic Solids Chemistry LibreTexts Does Boron Not Form Ionic Compounds Boron is found in nature on earth almost entirely as various oxides of b (iii), often associated with other elements. with its high ionization energy, low electron affinity, low electronegativity, and small size, however, boron does not form a metallic. to say that boron forms only covalent compounds is an oversimplification. You won't get monatomic cations like the. Does Boron Not Form Ionic Compounds.